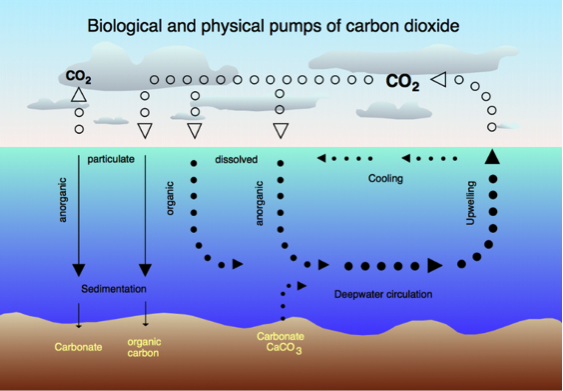
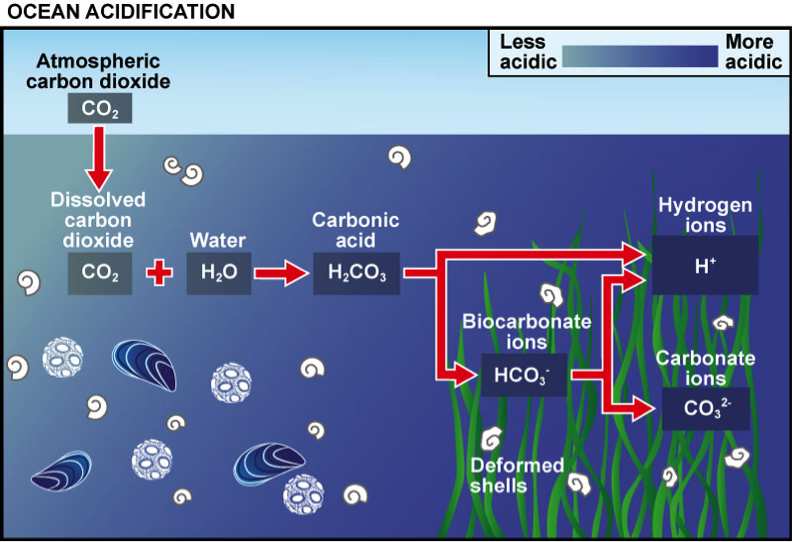
Carbon Cycle and Acidification:
The first principle we need to know in order to understand the fundamentals of Ocean Acidification is the carbon cycle. As shown in the picture, through Carbon cycle, CO2 is transferred between Oceans, terrestrial biosphere, lithosphere, and atmosphere. Once Carbon Dioxide is dissolved into the ocean water, it reacts with water, forming CO2, H2CO3, CO32-, and so on. Of course the concentration of each species differ depending on the seawater temperature, pressure, and alkalinity. However, the important point here is that through this cycle, Hydrogen ions are released into ocean, causing the acidification. Equations are as follows;
Acidification Rates:

The rates of acidification is actually much faster than it was predicted. Since the beginning of the industrial era, the ocean absorbed 525 billion tons of CO2, and presently around 22 million tons per day. According to the UK Ocean Acidification Research Programme, ocean pH already has decreased by about 30 %, and if we continue emitting CO2 at the same rate, ocean acidity will increase by about 150% by 2100, which we never have reached for past 400,000 years. The graph below shows changes in atmospheric and seawater CO2 and the seawater pH. It is obvious that the rates are accelerated, and as the atmospheric CO2 increases, that of seawater also increases, while pH decreases. At first, scientists didn’t worry about this process, because they thought a lot of chemicals (mostly ions) that were previously dissolved in the rivers and lakes will buffer the chemicals formed by dissolved CO2. However, the rates of this phenomenon was so fast, that natural buffering cannot keep up. The picture below is provided by Wikipedia, and it shows the changes in ocean water pH throughout the world. Almost every part is shown with green to yellowish colour which is -0.06 ~-0.1, definitely proves the ocean acidification.
Theoretical Effects on Marine Systems:
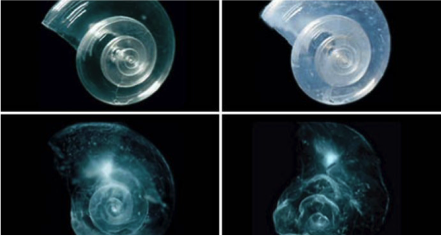
This phenomenon was so rapid that it didn’t give much time for marine ecosystems to adapt to it. Especially the ones with shells had their shells damaged by increasing acidity of the ocean water. The picture below is the result of the laboratory system.
The image to the left is a sea butterfly shell placed in seawater with increased acidity. The shell slowly dissolves over 45 days, showing that that shells are destroyed by the acidity of ocean. However, this is just one effect of the ocean acidification. Since the biological effects are more difficult to predict, than the chemical ones, it might be more dramatic than what we expect.
The image to the left is a sea butterfly shell placed in seawater with increased acidity. The shell slowly dissolves over 45 days, showing that that shells are destroyed by the acidity of ocean. However, this is just one effect of the ocean acidification. Since the biological effects are more difficult to predict, than the chemical ones, it might be more dramatic than what we expect.